How to write a research news release
This guide will help you get from that blank page to a first draft more quickly. It covers the main elements to include in a research news release, and how to structure them.
Contact media.relations@ubc.ca or 604.822.6397. Download the PDF version here.
Write it like a news story
The goal in writing a news release is to make it easy for newsroom decision-makers to imagine your item as a news story. The best way to do this is to write it like a news story.
What does this mean? Primarily, it means discarding academic jargon in favour of a conversational tone and everyday language.
If you use a term that would be unfamiliar to a lay reader, define it: “A planet’s global magnetic field arises from what scientists call a dynamo: a flow of molten metal within the planet’s core that produces an electrical current.”
It also means identifying and highlighting the most newsworthy elements. For this, it may be helpful to review our list of traditional news values:
- Timeliness
- Impact
- Disaster/threat
- Conflict
- Discovery
- Superlatives
- Human interest
- Proximity
- Oddity
- Prominence
Structure it like a news story
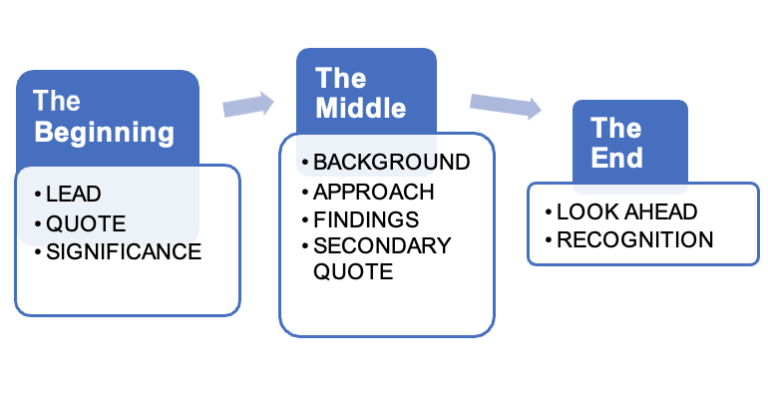
Structure your news release like a news story. Like all stories, it has a beginning, middle, and end.
You will find some news releases that depart from this formula. Experienced writers know the rules, and when to break them.
You don’t have to stick to this formula, but if you do, it will be hard to go wrong.
The ideal length for a news release is 500-600 words. You don’t have to tell the story comprehensively, because you will link to the research paper and include a contact for media who want to learn more.
Below is the basic structure of a research news release.
How story structure works
The Beginning covers the news, why it matters, and a quote.
The Middle provides background and context for the study and describes how it was conducted. It covers the findings in more detail.
The End looks to the future, suggesting where the discovery could lead and takes care of any acknowledgements that are owed
BEGINNING |
|
| Lead | The single most newsworthy thing people need to know |
| Lead quote | Researcher sums it all up in a punchy sentence or two |
| Significance | Why it matters |
MIDDLE |
|
| Background | Provide context and/or set up the problem |
| Approach | How they went about the study; methodology |
| Findings | Details about main finding and secondary findings of note, with stats and data |
| Secondary quote(s) | Researcher elaborates or provides another interesting observation |
END |
|
| Lookahead | Next steps; what has to happen; new questions or avenues of research |
| Credit | Acknowledge partners, granting agencies, etc. |
| Credit should include a reference and link to the journal publication, if it hasn’t already been mentioned. | |
Examples of research news releases
Example 1
| Headline | How to make carbon pricing palatable to air travellers |
| Lead quote | “People have the perception that the oil companies are the ones responsible for climate change, or at least more responsible than they are,” says study co-author David Hardisty, an assistant professor of marketing and behavioural science at UBC Sauder School of Business. “Consumers are more supportive of carbon pricing if it’s directed at the fossil fuel producers and importers than if it’s directed at consumers.” |
| Significance | The findings, newly published in the Journal of Environmental Psychology, suggest a path forward for the global airline industry which is a significant and growing source of carbon emissions. In the absence of policy changes, emissions from air travel are on track to triple over the coming decades |
| Background |
Carbon offsets could mitigate this. These are fees paid by polluters which are then redirected toward projects that reduce emissions elsewhere in the world. The airline industry’s negative impact on the environment could be partly addressed by routinely packaging carbon offsets along with the cost of tickets. But would consumers go for it? That’s what the researchers aimed to find out. The study, conducted in partnership with the Environmental Defense Fund, consisted of two separate online surveys of more than 1,800 U.S. participants. |
| Approach | “We wanted to gauge consumers’ reaction to a $14 carbon fee that was presented to them in several different ways at the time of a hypothetical ticket purchase,” explains study co-author Alec Beall, a postdoctoral researcher in UBC’s psychology department. “The dollar amount of the charge was the same, but it was described as either a ‘carbon offset’ or a ‘tax,’ for either ‘aviation fuel production and import’ or ‘airplane travel.’” |
| Findings |
The words mattered. Consumers were consistently more likely to choose a flight that included a carbon price when it was described as a “carbon offset for aviation fuel production and import” instead of a “carbon tax for airplane travel.” Importantly, consumers even chose more expensive tickets with a fee described this way over cheaper tickets that had no extra $14 fee attached at all. |
| Secondary quote |
“Taxes feel like you’re charging people money for nothing,” said Hardisty. “Whereas an offset is the idea that, ‘Sure we’re paying, but we kind of have an idea where that payment is going, to make the environment better,’ which is what people want.” Once consumers understand that a flight costs more because its price includes a carbon offset, they are likely to choose that flight and pay their part of the offset. |
| Lookahead | The study offers a blueprint to the air industry for garnering consumer support for carbon pricing: make it clear to consumers that their money is helping address the causes and consequences of carbon emissions. |
Example 2
| Headline | Sepsis leading cause of death worldwide |
| Lead | New research published today in The Lancet has found that sepsis is responsible for the most deaths worldwide, even more than cancer or coronary disease — previously believed to be the leading causes of death globally. |
| Significance | The study determined that, in 2017, 48.9 million cases of sepsis were diagnosed worldwide and 11 million people died of the condition. This represents one in five deaths worldwide — twice as many as previously estimated. |
| Lead quote |
“Eleven million deaths worldwide is a tragedy, especially as most cases of sepsis are either preventable or treatable if caught in time,” said study co-author Dr. Tex Kissoon, a professor at UBC Faculty of Medicine’s department of pediatrics and executive medical director of Global Health for Women and Children at BC Children’s Hospital. “We urgently need a coordinated global effort to tackle this crisis,” added Kissoon. “Raising awareness of sepsis among both patients and medical practitioners and ensuring everyone has access to public health measures to prevent and treat sepsis can greatly reduce the burden and ravages of this condition.” |
| Background | Sepsis occurs when the body’s response to infection begins to damage its own tissues and organs. The condition can be fatal if not recognized and treated promptly. It can be particularly dangerous for young children, as 40 per cent of cases worldwide occur in children under five years old. |
| Approach | For the study, Kissoon and his colleagues examined data from more than 100 million deaths in 195 countries between the years 1990 and 2017. Previous estimates had been based on adult hospital admissions from only seven high-income countries. |
| Secondary quote | “The impact of sepsis on low- and middle-income countries was largely a black hole before,” said Kissoon. “For too long, global sepsis numbers had simply been extrapolated from a handful of high- income countries. It took years of work to obtain more complete data in nearly every country in the world to get a much more robust picture of the global burden of sepsis. We found that it is a much bigger global threat than we thought.” |
| Findings | Although sepsis is a more significant threat than previously realized, Kissoon said it is promising that the incidence of cases is declining. In 1990, there were an estimated 60.2 million sepsis cases and 15.7 million deaths. By 2017, the incidence of sepsis had dropped by 19 per cent and deaths had dropped by 30 per cent. |
| Lookahead | “The biggest reasons for the decline in sepsis are advances in vaccination, access to clean drinking water and better nutrition, and housing due to the fact that nearly a billion people have been lifted out of poverty,” he said. “But there is still much work to do to address the nearly 49 million people that are still being affected by sepsis.” |
| Credit | The study was led by researchers at the Institute of Health Metrics and Evaluation at the University of Washington in Seattle. Kissoon is the only Canadian researcher involved in the international study. |
Example 3
| Headline | Huntington drug successfully lowers levels of disease-causing protein |
| Lead | An international clinical trial has found that a new drug for Huntington disease is safe, and that treatment with the drug successfully lowers levels of the abnormal protein that causes the debilitating disease in patients. |
| Significance | In the study published today in the New England Journal of Medicine, researchers from UBC and their colleagues have demonstrated for the first time that the drug, IONIS-HTTRX (now known as RO7234292) successfully lowers levels of the mutant huntingtin protein—the toxic protein that causes Huntington disease—in the central nervous system of patients. |
| Lead Quote | “This is a tremendously exciting and promising result for patients and families affected by this devastating genetic brain disorder,” said Dr. Blair Leavitt, neurologist and director of research at the Centre for Huntington Disease at UBC. “For the first time, we have evidence that a treatment can not only decrease levels of the toxic disease-causing protein in patients, but that it is also safe and very well tolerated.” |
| Background |
Leavitt, who is also a senior scientist at the Centre for Molecular Medicine and Therapeutics in the UBC faculty of medicine, treated all the Canadian participants in this study, including the first patient enrolled in the study in September 2015.
Huntington disease (HD) is a fatal genetic neurological disease. It usually develops in adulthood and causes abnormal involuntary movements, psychiatric symptoms and dementia. About one in 10,000 people in Canada have HD. To date, no effective treatments have been proven to slow down progression of this disorder. HD is caused by a single known genetic mutation, and each child of a carrier of the mutation has a 50 per cent chance of inheriting the disease. |
| Approach | The trial enrolled 46 patients with early HD at nine study centres in Canada, the United Kingdom, and Germany. Of the 46 patients, 34 were randomized to receive the drug and 12 were randomized to receive placebo. Each participant received four doses of the drug and all study participants completed the study and have continued to receive the active drug in an ongoing open-label study. The drug was administered monthly to patients via an injection directly into the cerebrospinal fluid. |
| Findings | The researchers, led by Dr. Sarah Tabrizi, director of the Huntington Disease Centre at University College London and global chief investigator of the IONIS-HTTRX clinical trial, found that the drug produced significant decreases in the levels of mutant huntingtin protein in the patients’ cerebrospinal fluid. No study participants experienced serious adverse reactions, suggesting that the treatment is safe and well tolerated by patients. |
| Lookahead | The drug is currently being evaluated in a large, phase-three multi-centre clinical trial being performed at the Centre for Huntington Disease at UBC and other HD centres around the world. This study is designed to determine whether the treatment slows or halts the progression of the disease symptoms. |
| Credit | It was sponsored by Ionis Pharmaceuticals and their partner Hoffmann-La Roche. |